Automotive batteries are essential components in vehicles, providing the electrical energy required to start the engine and power various electrical systems. Different types of automotive batteries vary in their design, function, and application based on the specific needs of the vehicle. Here are the most common types of automotive batteries and how they differ:
1. Lead-Acid Batteries
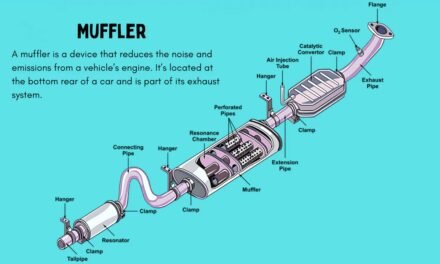
- Function: Lead-acid batteries are the most common type used in traditional internal combustion engine (ICE) vehicles. They are used to start the engine and provide power to electrical components like lights, radio, and air conditioning.
- Design: Composed of lead plates submerged in a sulfuric acid electrolyte solution. When the battery discharges, a chemical reaction occurs between the lead plates and the sulfuric acid, producing electricity.
- Types:
- Flooded Lead-Acid (FLA): The most basic form of lead-acid batteries, where the electrolyte is liquid and the plates are submerged in it. Requires periodic maintenance.
- Absorbent Glass Mat (AGM): A more advanced version of lead-acid batteries with fiberglass mats that absorb the electrolyte. AGM batteries are more durable, require no maintenance, and have better resistance to vibration.
- Gel Batteries: These use a gel electrolyte instead of liquid. Gel batteries are more resistant to deep discharge and are often used in more specialized applications, like in off-road vehicles or recreational vehicles.
- Applications: Lead-acid batteries are commonly used in passenger vehicles, commercial vehicles, and heavy-duty trucks.
- Advantages:
- Cost-effective
- Reliable
- Widely available
- Disadvantages:
- Heavier and bulkier compared to other battery types
- Shorter lifespan than newer technologies
- Less energy density
2. Lithium-Ion (Li-ion) Batteries
- Function: Lithium-ion batteries are commonly used in electric vehicles (EVs) and hybrid vehicles due to their higher energy density, lighter weight, and longer lifespan. They are also used in high-performance vehicles, including sports cars and motorcycles.
- Design: Composed of lithium metal oxide and graphite anodes with a liquid electrolyte. Lithium-ion batteries store energy by shuttling lithium ions between the anode and cathode.
- Applications: Electric vehicles (EVs), hybrid vehicles (HEVs and PHEVs), motorcycles, and power tools.
- Advantages:
- Higher energy density, which allows for a longer driving range in EVs
- Lighter and smaller than lead-acid batteries
- Longer lifespan (typically 8–15 years in EVs)
- Faster charging times
- Disadvantages:
- More expensive than lead-acid batteries
- Sensitive to high temperatures and extreme conditions, which can shorten lifespan
- Requires advanced battery management systems (BMS) to regulate charging and discharging
3. Nickel-Metal Hydride (NiMH) Batteries
- Function: Nickel-metal hydride (NiMH) batteries are used primarily in hybrid electric vehicles (HEVs) as part of their powertrain system. They are less common in fully electric vehicles but still provide a reliable and relatively affordable energy source for hybrids.
- Design: NiMH batteries consist of a positive electrode made of nickel oxyhydroxide and a negative electrode made of hydrogen-absorbing alloys. They use a potassium hydroxide electrolyte.
- Applications: Used in hybrid vehicles (HEVs) like the Toyota Prius, Honda Insight, and other hybrids.
- Advantages:
- Safer than lithium-ion batteries because they are less flammable
- Less expensive than lithium-ion batteries
- Good energy efficiency and moderate energy density
- Better performance in cold temperatures compared to Li-ion batteries
- Disadvantages:
- Heavier and bulkier than lithium-ion batteries
- Shorter lifespan and less energy density than lithium-ion batteries
- Less efficient than lithium-ion in fully electric vehicles
4. Solid-State Batteries
- Function: Solid-state batteries are an emerging technology that could potentially replace liquid electrolyte batteries like lithium-ion in the future. They use a solid electrolyte rather than a liquid one to store and transfer energy.
- Design: Solid-state batteries are made up of a solid electrolyte (such as ceramic or polymer), lithium metal or a lithium alloy, and a solid electrolyte separator.
- Applications: While they are not yet widely used in production vehicles, they are being researched for future applications in electric vehicles and consumer electronics.
- Advantages:
- Higher energy density compared to liquid lithium-ion batteries
- Safer with a lower risk of fires or leaks
- Faster charging times
- Longer lifespan
- Disadvantages:
- Expensive to manufacture
- Still in the development stage for automotive use and not widely available
- Manufacturing challenges related to scalability
5. Zinc-Air Batteries
- Function: Zinc-air batteries are a type of metal-air battery that uses zinc as the anode and oxygen from the air as the cathode. These batteries are most commonly used in hearing aids, small electronics, and some electric vehicle prototypes.
- Design: Zinc-air batteries use a zinc anode, a separator that allows oxygen to enter from the surrounding air, and an electrolyte that allows the chemical reaction between zinc and oxygen to produce electricity.
- Applications: Electric vehicles (in prototype stage), consumer electronics, hearing aids, medical devices.
- Advantages:
- High energy density and lightweight
- Environmental friendliness (uses non-toxic materials like zinc and oxygen)
- Potential for cheaper production costs
- Disadvantages:
- Shorter lifespan compared to lithium-ion and lead-acid batteries
- Limited rechargeability (often designed for single-use or limited cycles)
6. Supercapacitors (Ultracapacitors)
- Function: Supercapacitors are used in vehicles for energy storage in applications requiring quick bursts of power, such as hybrid vehicles or electric vehicle (EV) acceleration. They are sometimes used in combination with other batteries to enhance performance.
- Design: Supercapacitors store energy electrostatically, using two electrodes separated by an electrolyte. Unlike traditional batteries, they don’t rely on chemical reactions to store energy, so they can charge and discharge quickly.
- Applications: Hybrid vehicles, electric vehicles, energy recovery systems in race cars, and fast-charging devices.
- Advantages:
- Fast charging and discharging
- Long lifespan with virtually no degradation
- Can withstand a large number of charge cycles
- Disadvantages:
- Low energy density compared to batteries (cannot store as much energy)
- Expensive relative to other battery types
- Often used in combination with other energy storage systems
7. Flow Batteries
- Function: Flow batteries are an experimental technology with potential applications in electric vehicles, especially for heavy-duty and long-range vehicles. They store energy in liquid electrolyte solutions and use a pump to circulate the liquid through an electrochemical cell.
- Design: Flow batteries use two electrolyte solutions that are stored in external tanks and pumped through a cell stack to generate power. Energy is stored in the liquid electrolyte and is replenished or replaced as needed.
- Applications: Primarily experimental and used for large-scale storage or industrial applications. There is potential for EVs in the future, but they are not yet commercially viable for mainstream use.
- Advantages:
- Can provide large amounts of energy with minimal degradation over time
- Scalable for larger vehicles or longer ranges
- Can be quickly recharged by swapping electrolyte tanks
- Disadvantages:
- Lower energy density than lithium-ion batteries
- Bulky and complex design
- Expensive to produce and maintain
Conclusion
Automotive batteries come in various types, each with distinct characteristics suited to different applications. Lead-acid batteries are widely used in traditional vehicles, while lithium-ion batteries dominate the electric vehicle (EV) market due to their higher energy density and longer lifespan. Other battery technologies, like nickel-metal hydride (NiMH), solid-state batteries, and supercapacitors, offer benefits for hybrid vehicles, energy storage, and future EV developments. Each type of battery has its advantages and challenges, and their application will depend on factors like vehicle type, energy requirements, cost, and environmental considerations.
Hashtags
#AutomotiveBatteries #LeadAcidBattery #LithiumIonBattery #AGMBattery #GelCellBattery #NickelMetalHydrideBattery #EFBattery #AbsorbentGlassMat #TraditionalCarBattery #StartingLightingIgnition #LeadAcidBatteryUses #AffordableCarBatteries #EVBatteries #HighEnergyDensityBatteries #LiIonBatteriesForCars #LightweightBatteriesForEVs #AGMStartStopSystem #HighPerformanceCarBattery #AGMCarBatteryAdvantages #GelBatteriesForCars #SealedCarBatteries #BatteryForExtremeConditions #NiMHBatteriesForHybrids #NiMHBatteryTechnology #EFBStartStopSystem #EFBHighDurabilityBatteries #BatteryForVehicleStarting #CarBatteryForPowerStorage #ElectricVehicleBattery #BatteryForStartStopSystem #HybridVehicleBattery #EVPowerStorage